MDR and Traceability
– What Does It Mean for You as a Buyer??
Medical devices are subject to one of the most comprehensive regulatory frameworks in the EU: the Medical Device Regulation (MDR).
For buyers, procurement professionals and healthcare managers, this means both new requirements and increased assurance.
What is MDR?
MDR (EU 2017/745) is the EU regulation governing medical devices. Its purpose is to:
- Improve patient safety
- Ensure product quality
- Strengthen traceability throughout the entire product lifecycle
- Improve clinical documentation
The regulation sets higher requirements than previous directives, both for manufacturers and healthcare providers.

What Does Traceability Mean?
Traceability means that a product can be tracked from raw materials through production to delivery, often down to a specific batch or serial number.
This is essential in situations such as:
- Non-conformities
- Product recalls
- Quality follow-up
- Risk management
For buyers, this means greater transparency and better control.
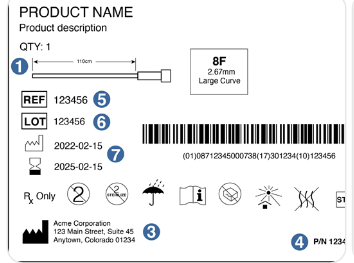
What Should You Verify During Procurement?
When procuring medical devices, it is important to ensure that the supplier can provide:
✔ CE marking according to MDR
✔ Technical documentation
✔ Clinical evaluation
✔ A risk management process
✔ A quality management system (e.g. ISO 13485)
✔ Clear batch and product identification
It is not only about the product itself, but about the entire quality system behind it.
Single-Use Products and Safety
For single-use products, traceability is particularly important because they:
- are used directly on patients
- are not reused or reprocessed
- must be safe at the point of delivery
A robust quality process at the manufacturer reduces the risk of deviations in the clinical environment.

Our Approach
At Cetromedical, we work systematically with quality and traceability throughout the entire product lifecycle through:
- documented processes
- controls at every stage of production
- clear batch traceability
- continuous risk management
For us, MDR is not just a regulatory requirement – it is part of our responsibility towards healthcare.
Summary
MDR brings higher requirements, but also higher levels of safety.
For buyers, it is essential to choose suppliers who actively work with quality, documentation and traceability.
Because patient safety begins long before a product reaches the examination room.
